Link to Virology explainers, Virologists and to the outer, more accessible edges of the science. Assembled and organized for my own use. If these help you, great..
NB: I haven't written out their full names, titles, or affiliations. Assume they've PhDs and more. There's no point in quoting randos or facebook foolishness.
Ed Rybicki @edrybicki
Björn Meyer @_b_meyer
Trevor Bedford @trvrb Bedford Lab @fredhutch.org
Isabella Eckerle, MD @EckerleIsabella
Dr. Zoë Hyde @DrZoeHyde
Dr. Krutika Kuppalli @KrutikaKuppalli
Ian M. Mackay, PhD @MackayIM
Dr. Angela Rasmussen @angie_rasmussen
Dr Emma Hodcraft @firefoxx66
CoVariants CoVariants.org
Prof. Akiko Iwasaki @VirusesImmunity
Marion Koopmans @MarionKoopmans
Brian Wasik @BrianRWasik
Lauring Lab @LauringLab
Bloom Lab @jbloom_lab
Tyler Starr @tylernstarr
Marija Backovic @marija_backovic
Dr. Arinjay Banerjee @sci_questions
DIY? SEC-MALS Validated SARS-CoV-2 Prefusion Trimeric Spike Variant Proteins
10/8/2021 How antiviral pill molnupiravir shot ahead in the COVID drug hunt, by Cassandra Willyard, Nature
Molnupiravir, like remdesivir, is a nucleoside analogue, which means it mimics some of the building blocks of RNA. But the compounds work in entirely different ways. When SARS-CoV-2 enters a cell, the virus needs to duplicate its RNA genome to form new viruses. Remdesivir is a ‘chain terminator’. It stops the enzyme that builds these RNA ‘chains’ from adding further links. Molnupiravir, on the other hand, gets incorporated into burgeoning RNA strands and, once inside, wreaks havoc. The compound can shift its configuration, sometimes mimicking the nucleoside cytidine and sometimes mimicking uridine. Those RNA strands become faulty blueprints for the next round of viral genomes. Anywhere the compound gets inserted and that conformational shift happens, a point mutation occurs, Plemper says. When enough mutations accumulate, the viral population collapses. “That is what we term lethal mutagenesis,” he adds. “The virus essentially mutates itself to death.” And because the mutations accumulate randomly, it’s difficult for viruses to evolve resistance to molnupiravir — another plus for the compound.
Whether this clinical-trial success story will translate into a global game-changer in the fight against the pandemic isn’t yet clear. This week, two Indian drugmakers testing molnupiravir in people with moderate illness due to COVID-19 sought to end their trials because they saw no “significant efficacy” for the experimental drug. Also, Merck’s findings, which apply to people with mild-to-moderate cases of COVID-19, were disclosed only in a press release — and have yet to be pored over by scientists and submitted to regulators for approval
The United States has agreed to purchase 1.7 million courses of molnupiravir for US$1.2 billion, which works out to about $700 per 5-day course. That’s far less than the price of remdesivir or monoclonal antibodies, but still too costly for much of the world. Merck, which is co-developing the compound with Ridgeback, has struck licensing agreements with five Indian manufacturers of generic drugs. Those deals allow the manufacturers to set their own price in India and 100 other low- and lower-middle-income countries.
9/30/2021
9/28/2021 A prenylated dsRNA sensor protects against severe COVID-19, Science, for list of 50+ authors, follow link
Inherited genetic factors can influence the severity of COVID-19, but the molecular explanation underpinning a genetic association is often unclear. Intracellular antiviral defenses can inhibit the replication of viruses and reduce disease severity. To better understand the antiviral defenses relevant to COVID-19, we used interferon-stimulated gene (ISG) expression screening to reveal that OAS1, through RNase L, potently inhibits SARS-CoV-2. We show that a common splice-acceptor SNP (Rs10774671) governs whether people express prenylated OAS1 isoforms that are membrane-associated and sense specific regions of SARS-CoV-2 RNAs, or only express cytosolic, nonprenylated OAS1 that does not efficiently detect SARS-CoV-2. Importantly, in hospitalized patients, expression of prenylated OAS1 was associated with protection from severe COVID-19, suggesting this antiviral defense is a major component of a protective antiviral response.
10/11/2021 A Neanderthal OAS1 isoform Protects Against COVID-19 Susceptibility and Severity: Results from Mendelian Randomization and Case-Control Studies, medRxIv
by Sirui Zhou, Guillaume Butler-Laporte, Tomoko Nakanishi, David Morrison, Jonathan Afilalo, Marc Afilalo, Laetitia Laurent, Maik Pietzner, Nicola Kerrison, Kaiqiong Zhao, Elsa Brunet-Ratnasingham, Danielle Henry, Nofar Kimchi, Zaman Afrasiabi, Nardin Rezk, Meriem Bouab, Louis Petitjean, Charlotte Guzman, Xiaoqing Xue, Chris Tselios, Branka Vulesevic, Olumide Adeleye, Tala Abdullah, Noor Almamlouk, Yiheng Chen, Michaël Chassé, Madeleine Durand, Michael Pollak, Clare Paterson, Hugo Zeberg, Johan Normark, Robert Frithiof, Miklós Lipcsey, Michael Hultström, Celia M T Greenwood, Claudia Langenberg, Elin Thysell, Vincent Mooser, Vincenzo Forgetta, Daniel E. Kaufmann, ichards
9/26/2021 Key Stages of the SARS-CoV-2 Life Cycle, Pfizer
SARS-CoV-2 proteolysis is driven by two virally encoded proteases, the 3C-like protease, 3CL (known as the main protease); and the papain-like protease.2,4 The 3CL protease cleaves the viral pp1ab polyprotein at 11 sites, while the papain-like protease cleaves at 3 sites, generating proteins critical for viral replication.2,4 Despite coronaviruses being subject to extensive mutagenesis, proteases are highly conserved among coronaviruses. SARS-CoV-2 proteases are potential targets for stopping replication.2,3,6,7
[ some brief animations, mainly ]
9/25/2021 Lack of Evidence of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Spillover in Free-Living Neotropical Non-Human Primates, Brazil, by Lívia Sacchetto 1,†OrcID,Bárbara Aparecida Chaves 2,3,4,†OrcID,Edson Rodrigues Costa 2OrcID,Aline Souza de Menezes Medeiros 2,Marcelo Gordo 5OrcID,Danielle Bastos Araújo 6,7,Danielle Bruna Leal Oliveira 6,7,8OrcID,Ana Paula Betaressi da Silva 9,Andréia Francesli Negri 9,Edison Luiz Durigon 6,10OrcID,Kathryn A. Hanley 11,Nikos Vasilakis 12,13,14,15,16OrcID,Marcus Vinícius Guimarães de Lacerda 2,3,17,* andMaurício Lacerda Nogueira 1,*OrcID, Viruses
Marion Koopmans: Twitter[ @MarionKoopmans ]: "Primates are sensitive to SARS COV 2 and in zoo’s have been infected. This study looked at free-living primates, found no evidence for reverse spillover."
9/24/2021 Caspase-4/11 exacerbates disease severity in SARS-CoV-2 infection by promoting inflammation and thrombosis, by Mostafa Eltobgy, Ashley Zani, Adam Kenney, Shady Estfanous, Eunsoo Kim, Asmaa Badr, Cierra Carafice, Kylene Daily, Owen Whitham, Maciej Pietrzak, Amy Webb, Jeffrey Kawahara, Adrian Eddy, Parker Denz, Mijia Lu, Mahesh KC, Mark Peeples, Jianrong Li, Jian Zhu, Jianwen Que, Richard Robinson, Oscar Rosas Mejia, Rachael Rayner, Luanne Hall-Stoodley, Stephanie Seveau, Mikhail A Gavrilin, Andrea Tedeschi, Santiago Partida-Sanchez, Frank Roberto, Emily Hemann, Eman Abdelrazik, Adriana Forero, Shahid Nimjee, Prosper Boyaka, Estelle Cormet-Boyaka, Jacob Yount, Amal Amer, bioRxiv
9/13/2021 Some thoughts on gain of function (GoF) and virology research in general, by Stuart Neil, PhD @stuartjdneil on twitter
One of these links takes you to a browser friendly threadreaderapp version of Neil's twitter thread. The other takes you to his twitter bio.
Credit: Janet Iwasa, University of Utah
2021/07/28 How the coronavirus infects cells — and why Delta is so dangerous, by Megan Scudellari, Nature
Scientists are unpicking the life cycle of SARS-CoV-2 and how the virus uses tricks to evade detection.
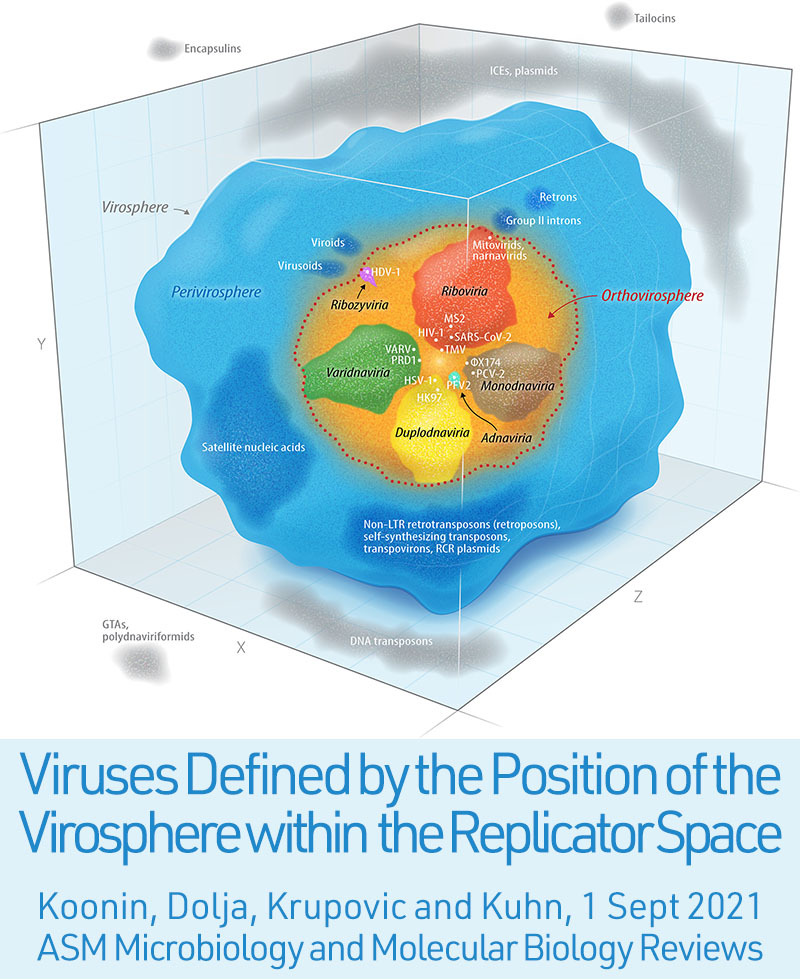
9/1/2021: "We argue that the virosphere proper, here termed orthovirosphere, consists of a distinct variety of replicators that encode structural proteins encasing the replicators’ genomes, thereby providing protection and facilitating transmission among hosts."
6/17/2021 The quest for a pill to fight viruses gets a $3.2 billion boost, by Carolyn Y. Johnson, The Washngton Post
“Investors are totally uninterested in antivirals. Even if you can demonstrate you can make a couple billion dollars, nobody cares,” said Ann Kwong, a virologist who played a leading role in developing an antiviral approved against hepatitis C at Vertex Pharmaceuticals, along with an influenza treatment. “What they really want is a chronic treatment. Nobody ever gets cured of high cholesterol.”
4/29/2021 Hopes rise for coronavirus drug remdesivir, by Heidi Ledford, Nature
Despite conflicting studies, results from largest trial yet show the antiviral speeds up recovery, putting it on track to become a standard of care in the United States.
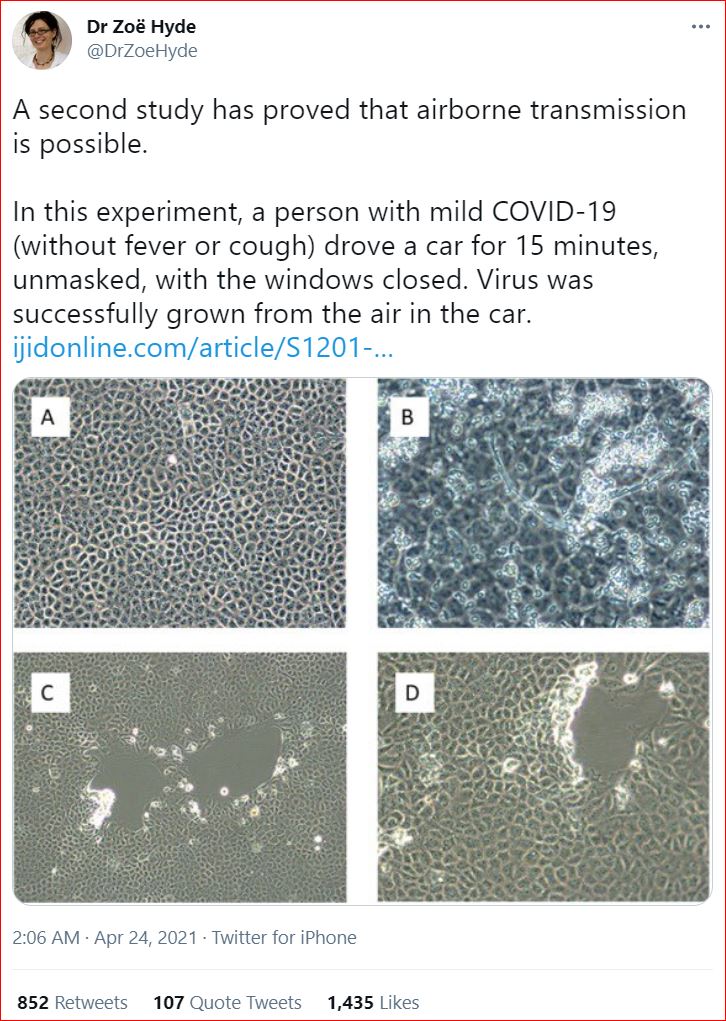
4/23/2021 Isolation of SARS-CoV-2 from the air in a car driven by a COVID patient with mild illness, by John A. Lednicky, Michael Lauzardo Md., M. Alam, Maha A. Elbadry, Caroline J. Stephenson, Julia C. Gibson, J. Glenn Morris Jr., International Journal of Infectious Diseases
- SARS-CoV-2 was isolated from air in a car driven by a COVID patient.
- Virus was cultured from samples with a particle range of 0.25 to 0.50 μm.
- A substantial component of transmission risk appears to be via aerosolized virus.
Methods
We used a Sioutas personal cascade impactor sampler (PCIS) to screen for SARS-CoV-2 in a car driven by a COVID-19 patient. The patient, who had only mild illness without fever or cough and was not wearing a mask, drove the car for 15 minutes with the air conditioning turned on and windows closed. The PCIS was clipped to the sun-visor above the front passenger seat and was retrieved from the car two hours after completion of the drive.
ResultsSARS-CoV-2 was detectable at all PCIS stages by PCR and was cultured from the section of the sampler collecting particles in the 0.25 to 0.50 μm size range.
Conclusions
Our data highlight the potential risk of SARS-CoV-2 transmission by minimally symptomatic persons in the closed space inside of a car and suggest that a substantial component of that risk is via aerosolized virus.
4/22/2020 Coronavirus: the first three months as it happened
Nature recorded major events as the pandemic spread across the globe.
4/14/2021 The race for antiviral drugs to beat COVID — and the next pandemic, by Elie Dolgin, Nature
Despite dire warnings, a stockpile of ready compounds to fight viral pandemics was sorely lacking. Can drugmakers finally do the right thing?
3/11/2021 mRNA vaccine protects monkeys against HIV-like virus, by Liz Highleyman, nam/aidsmap
The same messenger RNA (mRNA) approach used for the highly effective Pfizer-BioNTech and Moderna COVID-19 vaccines also shows promise for protection against HIV, according to a presentation this week at the virtual Conference on Retroviruses and Opportunistic Infections.
Despite more than three decades of research, scientists have had little success developing vaccines to prevent HIV. To date, only one vaccine regimen – a canarypox vector primer followed by a gp120 protein booster – has demonstrated partial protection in human studies; however, it was not effective in a recent large trial.
Dr Peng Zhang of the US National Institute of Allergy and Infectious Diseases (NIAID) and colleagues, in collaboration with Moderna, are taking a different approach. The investigators designed a vaccine regimen that introduces mRNA into a cell and instructs it to make the outer, envelope proteins of three different subtypes of HIV, plus the structural gag protein from SIV, HIV's simian cousin. The cells then assemble these proteins to make virus-like particles that trigger an immune response. These particles are not viruses because they have no genetic material of their own and cannot reproduce – but they look like viruses to the immune system.
3/2/2021 Study looks at the impact of Stabilising SARS-Cov-2 Spike through “PP” mutations and Cleavage site removal. Combination of PP and Cleavage site removal (as seen in J&J/Novavax vaccine) was beneficial, “PP” mutation alone (as seen in Pfizer/Moderna) was not, COVID Data Review
3/2/2021 Introduction of Two Prolines and Removal of the Polybasic Cleavage Site Lead to Higher Efficacy of a Recombinant Spike-Based SARS-CoV-2 Vaccine in the Mouse Model, by Fatima Amanat, Shirin Strohmeier, Raveen Rathnasinghe, Michael Schotsaert, Lynda Coughlan, Adolfo García-Sastre, Florian Krammer, American Society for Microbiology
3/2/2021 SARS-CoV-2 RBD-Tetanus toxoid conjugate vaccine induces a strong neutralizing immunity in preclinical studies, [by dozens of scientists], bioRxiv
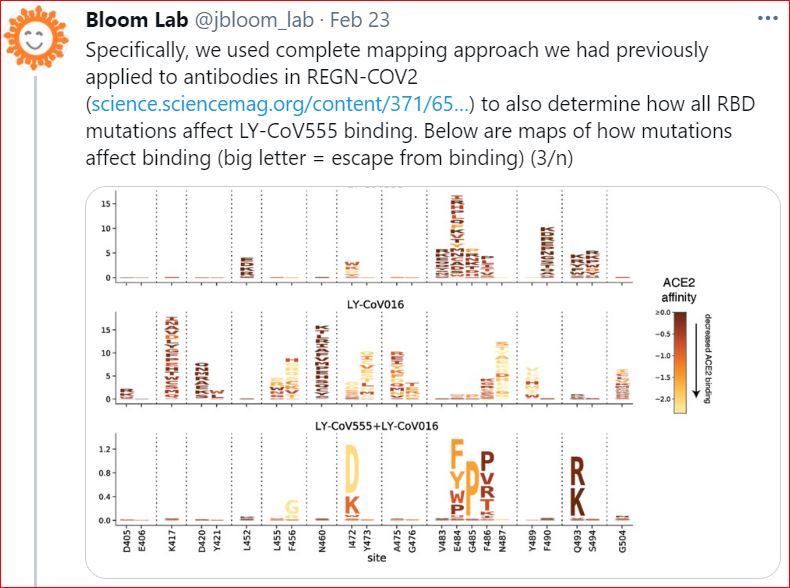
2/23/2021 "We have completely mapped #SARS_CoV_2 mutations that escape binding by LY-CoV555 (antibody that forms the basis for Eli Lilly's bamlanivimab) both alone & in cocktail with LY-CoV016 in a new study," Bloom Lab
2/22/2021 Complete map of SARS-CoV-2 RBD mutations that escape the monoclonal antibody LY-CoV555 and its cocktail with LY-CoV016, by Tyler N. Starr, Allison J. Greaney, Adam S. Dingens, Jesse D. Bloom, bioRxiv
The dynamics of adaptation on correlated fitness landscapes, by Sergey Kryazhimskiya, Gašper Tkacikˇ and Joshua B. Plotkina, Proceedings of the National Academy of Sciences of the United States of America
2021-01-31 NYC Reveals White New Yorkers Have Received Lion's Share Of COVID-19 Vaccine Doses, by Sydney Pereira, Jake Dobkin and Nsikan Akpan, the Gothamist
Three white residents receive a COVID-19 vaccine for every Black or Latino person in the city, according to new demographic data released by the mayor’s office on Sunday.
2021-01-29 Virus expert Trevor Bedford warns of "convergent evolution" amid COVID-19 mutations, Margaret Brennan, Face the Nation
2021-01-27 We Cannot Rely Exclusively on Vaccines to End the Pandemic, Angela Rasmussen, Slate
2021/01/21 mRNA Vaccines: What Happens, Derek Lowe, Sciencemag
2021/01/02 Putting PCR into real-time, Ian M Mackay, PhD, Virology Down Under
2020/12/28 The mechanics of the polymerase chain reaction (PCR)…a primer, Ian M Mackay, PhD
2020/10/24 The Swiss Cheese Respiratory Virus Pandemic Defence, Mackay
2020/10/23 Virology, transmission, and pathogenesis of SARS-CoV-2, Cevik, Kuppalli and Kindrachuk, BMJ
2020/05/15 The sprint to solve coronavirus protein structures — and disarm them with drugs, by Megan Scudellari,
Lying in bed on the night of 10 January, scrolling through news on his smartphone, Andrew Mesecar got an alert. He sat up. It was here. The complete genome of a coronavirus causing a cluster of pneumonia-like cases in Wuhan, China, had just been posted online.
2020/05/14 Dozens of coronavirus drugs are in development — what happens next? by Heidi Ledford, Nature
2020/05/04 Profile of a killer: the complex biology powering the coronavirus pandemic, by David Cyranoski, Nature
In 1912, German veterinarians puzzled over the case of a feverish cat with an enormously swollen belly. That is now thought to be the first reported example of the debilitating power of a coronavirus. Veterinarians didn’t know it at the time, but coronaviruses were also giving chickens bronchitis, and pigs an intestinal disease that killed almost every piglet under two weeks old.
The link between these pathogens remained hidden until the 1960s, when researchers in the United Kingdom and the United States isolated two viruses with crown-like structures causing common colds in humans. Scientists soon noticed that the viruses identified in sick animals had the same bristly structure, studded with spiky protein protrusions. Under electron microscopes, these viruses resembled the solar corona, which led researchers in 1968 to coin the term coronaviruses for the entire group.
Of the viruses that attack humans, coronaviruses are big. At 125 nanometres in diameter, they are also relatively large for the viruses that use RNA to replicate, the group that accounts for most newly emerging diseases. But coronaviruses really stand out for their genomes. With 30,000 genetic bases, coronaviruses have the largest genomes of all RNA viruses. Their genomes are more than three times as big as those of HIV and hepatitis C, and more than twice influenza’s.
2020/04/28 The race for coronavirus vaccines: a graphical guide, by Ewen Callaway, Nature
Eight ways in which scientists hope to provide immunity to SARS-CoV-2 .
2020/04/20 How does COVID-19 kill? Uncertainty is hampering doctors’ ability to choose treatments, by Heidi Ledford, Nature
Doctors are reaching for drugs that dampen the immune response — but these also undermine the body’s own fight against the coronavirus.
2020/04/07 Why measles deaths are surging — and coronavirus could make it worse, by Leslie Roberts, Nature
2020/02/18 When will the coronavirus outbreak peak? by David Cyranoski, Nature
On 11 February, Zhong Nanshan, a prominent Chinese physician leading a panel of experts helping to control the outbreak, said that the coronavirus will possibly peak by the end of February. Zhong, who is famous for discovering the SARS virus, said the situation had improved with government control measures, such as travel restrictions and extended holidays, although he admitted that it was still a “difficult period” for Wuhan.
Sebastian Funk, a statistician who models infectious diseases and who coauthored the analysis, says the prediction is based on an estimate that one infected person in Wuhan was, on average, infecting between 1.5 and 4.5 others — a measure known as the virus’s effective reproduction number, or R — before the travel restrictions were introduced on 23 January.
2020/02/15 More than 80 clinical trials launch to test coronavirus treatments, by Amy Maxmen, Nature
As HIV drugs, stem cells and traditional Chinese medicines vie for a chance to prove their worth, the World Health Organization attempts to bring order to the search.
2020/01/24 Wuhan scientists: What it’s like to be on lockdown, by David Cyranoski & Andrew Silver, Nature
Measures to contain a new virus’s spread have cut off the city's researchers.
2020/01/22 China coronavirus: Six questions scientists are asking, by Ewen Callaway & David Cyranoski, Nature
Researchers are racing to find out more about the epidemiology and genetic sequence of the coronavirus spreading in Asia and beyond.
2020/01/21 Stop the Wuhan virus, Nature
The virus has been spreading. On 21 January, as Nature went to press, there were almost 300 reported cases — seven times the figure stated five days earlier. Over the past week, authorities in South Korea, Thailand and Japan have also reported cases. Researchers at Imperial College London who have modelled the outbreak on the basis of estimates of travel out of Wuhan say the virus might have infected as many as 1,700 people.
The virus, which still lacks a formal name, is being called 2019-nCOV. It is a relative of both the deadly severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS) viruses. People with the virus report a fever along with other symptoms of lower-respiratory infection such as a cough or breathing difficulties. The first people infected in China are understood to have caught the virus in one of Wuhan’s live animal and seafood markets — probably from an animal. Some 95% of the total cases, including those in Japan, South Korea and Thailand, also involved people who had been to Wuhan.
2020/01/21 How quickly does the Wuhan virus spread? by Nicky Phillips , Smriti Mallapaty & David Cyranoski, Nature
Chinese officials have confirmed that the virus is spreading between people, but it’s still unclear how easily this happens.
2020/01/20 New virus surging in Asia rattles scientists, by David Cyranoski, Nature
Chinese officials reported more than 100 new infections and South Korea confirmed its first case.
2020/01/08 New virus identified as likely cause of mystery illness in China, by David Cyranoski, Nature
As of 5 January, 59 people in the east-central city of Wuhan have been infected with the mystery virus, with 7 in a critical condition, according to the Wuhan Municipal Health Commission.
Update: On 9 January, Chinese state media reported that scientists have identified a new coronavirus as the likely cause of a pneumonia-like illness that has sickened dozens of people. Researchers have sequenced the virus's genome, and fifteen patients have tested positive to the virus, according to Xinhua news agency.
2019/10/31 Measles erases immune ‘memory’ for other diseases, by Giorgia Guglielmi, Nature
Measles infections in children can wipe out the immune system’s memory of other illnesses such as influenza, according to a pair of studies[[1]] [[2]]. This can leave kids who recover from measles vulnerable to other pathogens that they might have been protected from before their bout with the virus.
The Science study is the first to show definitive evidence that measles can destroy this immune memory, says Harvard T. H. Chan School of Public Health in Boston, Massachusetts, infectious-disease immunologist Michael Mina.
2019/10/30 South Korea deploys snipers and drones to fend off deadly pig virus, by Mark Zastrow, Nature
Outbreaks of African swine fever have killed millions of pigs across Asia, and the virus has recently been detected in wild boars near the North Korean border.
2019/09/18 Nature Outlook: Influenza, Nature
2019/09/18 Towards a universal flu vaccine, by Michael Eisenstein, Nature
So far, antibodies have been the primary focus of the vaccine community because they represent a crucial first line of defence against circulating virus particles, but T cells provide critical protection by containing infection once it is under way. “People get exposed and infected every two or three years on average,” says Sarah Gilbert, who heads vaccine development at the University of Oxford’s Jenner Institute, UK. “The vast majority of these infections are either asymptomatic or mild,” she says, “and the reason is that people have a T-cell response that’s strong enough to protect them.”
2019/09/18 Flu on the farm, by Cassandra Willyard, Nature
Farms help to spread influenza but they might be an early warning system for the next human pandemic.
2019/04/25 Spread of deadly pig virus in China hastens vaccine research, by Smriti Mallapaty, Nature
The virus is not harmful to humans, but is taking a huge economic toll on the pork industry.
2018/11/01 Machine learning helps to identify the animals that deadly viruses call home, by Ewen Callaway, Nature
But the model will need further refining before it can pinpoint species-level disease reservoirs.
2018/11/01 Machine learning spots natural selection at work in human genome, by Amy Maxmen, Nature
Scientists are using artificial intelligence to identify genetic sequences molded by evolutionary pressures.
2018/03/19 Machine learning spots treasure trove of elusive viruses, by Amy Maxmen, Nature
In recent years, researchers have hunted for unknown viruses by sequencing DNA in samples taken from various environments. To identify the microbes present, researchers search for the genetic signatures of known viruses and bacteria — just as a word processor’s ‘find’ function highlights words containing particular letters in a document. But that method often fails, because virologists cannot search for what they do not know. A form of AI called machine learning gets around this problem because it can find emergent patterns in mountains of information. Machine-learning algorithms parse data, learn from them and then classify information autonomously.
“Previously, people had no method to study viruses well,” says Jie Ren, a computational biologist at the University of Southern California in Los Angeles. “But now we have tools to find them.”
2018/01/03 Deep learning sharpens views of cells and genes, by Amy Maxmen, Nature
Neural networks are making biological images easier to process.
2017/12/01 Bat cave solves mystery of deadly SARS virus — and suggests new outbreak could occur, by David Cyranoski, Nature
Chinese scientists find all the genetic building blocks of SARS in a single population of horseshoe bats.
2017/06/15 Bats are global reservoir for deadly coronaviruses, by Amy Maxmen, Nature
2017/04/05 Machine learning predicts the look of stem cells, by Amy Maxen, Nature
2017/02/23 Inside the Chinese lab poised to study world's most dangerous pathogens, by David Cyranoski, Nature
A laboratory in Wuhan is on the cusp of being cleared to work with the world’s most dangerous pathogens. The move is part of a plan to build between five and seven biosafety level-4 (BSL-4) labs across the Chinese mainland by 2025, and has generated much excitement, as well as some concerns.
The lab was certified as meeting the standards and criteria of BSL-4 by the China National Accreditation Service for Conformity Assessment (CNAS) in January. The CNAS examined the lab’s infrastructure, equipment and management, says a CNAS representative, paving the way for the Ministry of Health to give its approval.
BSL-4 is the highest level of biocontainment: its criteria include filtering air and treating water and waste before they leave the laboratory, and stipulating that researchers change clothes and shower before and after using lab facilities. Such labs are often controversial. The first BSL-4 lab in Japan was built in 1981, but operated with lower-risk pathogens until 2015, when safety concerns were finally overcome.
The expansion of BSL-4-lab networks in the United States and Europe over the past 15 years — with more than a dozen now in operation or under construction in each region — also met with resistance, including questions about the need for so many facilities.
2012/08/28 Pig fever sweeps across Russia, by Ewen Callaway, Nature
Scientists first encountered African swine fever in the 1920s in domestic pigs in Kenya, where the vicious haemorrhagic fever felled nearly every animal infected. The virus, which is also carried by warthogs and ticks without causing disease, is now endemic in much of sub-Saharan Africa, limiting pig farming there. It does not infect humans. In 1957, the virus jumped to Portugal after pigs near Lisbon’s airport were fed infected human food scraps (the virus particles can survive meat curing processes). It then hit Spain, and import of the region’s ham — including the coveted jamón ibérico — was banned by many countries, until the disease was eradicated in Spain and Portugal in the mid-1990s.